compound
What is a compound?
In chemistry, a compound is a substance made up of two or more different chemical elements combined in a fixed ratio. When the elements come together, they react with each other and form chemical bonds that are difficult to break. These bonds form as a result of sharing or exchanging electrons between atoms.
A compound is sometimes confused with a mixture, but the two differ in important ways. In a mixture, substances are combined, but there's no chemical reaction or bonding like that found in compounds. The individual substances in a mixture retain their own properties and can be separated into their original components. Air, saltwater and liquid dish soap are examples of mixtures.
In a compound, the different elements react with each other and form strong bonds that require significant energy to break apart. The elements don't retain their individual properties, nor does the compound necessarily behave like those elements. Water, table salt and baking soda are examples of compounds.
A compound is identified by its name and chemical formula. The formula reflects the compound's elemental makeup. It includes the chemical symbol for each element in the compound, and the number of atoms for that element if there's more than one. For instance, the chemical formula for sulfuric acid is H2SO4, which means the compound is made up of two hydrogen (H) atoms, one sulfur (S) atom, and four oxygen (O) atoms.
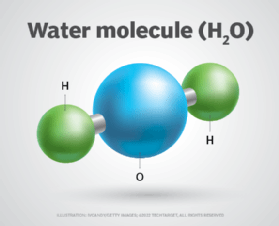
A compound often looks and behaves differently from its constituent elements. Consider a water (H2O) molecule, which is made up of two hydrogen atoms and one oxygen atom. Both hydrogen and oxygen are gases at room temperature and normal atmospheric pressure. But when they combine to form water, the chemistry changes and the resulting substance is a liquid at room temperature and normal atmospheric pressure.
The process of compound formation varies from one compound to the next. In some cases, the chemical reaction occurs slowly, such as when iron (Fe) and oxygen combine to form iron oxide (FeO). In other cases, the reaction happens quickly, as when lithium (Li) and oxygen combine to create lithium oxide (Li2O). Sometimes, a new compound might form when an element is exposed to an existing compound. For example, when pure elemental sodium (Na) is submerged into liquid water (H2O), a reaction occurs, resulting in the formation of sodium hydroxide (NaOH) and the release of hydrogen gas.
Types of compound bonds
Compounds are often categorized by the type of bond that holds them together. There are two primary types of bonds:
- Covalent bonds. Bonding occurs when two nonmetal atoms share pairs of electrons to form a stable bond. Examples of covalent compounds include water, carbon dioxide (CO2) and hydrogen chloride (HCl).
- Ionic bonds. Bonding occurs when a metal atom and a nonmetal atom exchange valence electrons to form a stable bond. Examples of ionic compounds include sodium chloride (NaCl), magnesium sulfate (MgSO4) and sodium bicarbonate (NaHCO3).
Not all compounds fit neatly into one category or the other. Some compounds, such as potassium Sulfate (K2SO4) and sodium hydroxide, contain both covalent and ionic bonds.
Compounds are sometimes distinguished by whether they're made up of molecules. A molecule is a structure that consists of two or more atoms that are chemically bonded together. The structure might be an elemental or compound molecule. An elemental molecule contains only one type of atom, such as ozone (O3) or chlorine (Cl2). A compound molecule is made up of one or more different elements. Covalent compounds are built on these types of molecules and are often referred to as molecular compounds.
There's no clear consensus on whether ionic compounds are molecular-based. Some sources consider them to have an underlying molecular structure, in which case, all compounds are molecular structures. Other sources treat the ionic compound as more of a lattice network than a series of molecules, differentiating them from covalent compounds and elementary molecules. Compounds that contain both covalent and ionic bonds bring even more uncertainly to this issue.
A small subset of elements -- helium, neon, argon, krypton, xenon and radon -- are known as noble or inert gases and don't readily bond with other elements to form compounds. However, elements such as oxygen, chlorine and fluorine readily combine with other elements to form compounds.
See also: table of physical constants.
